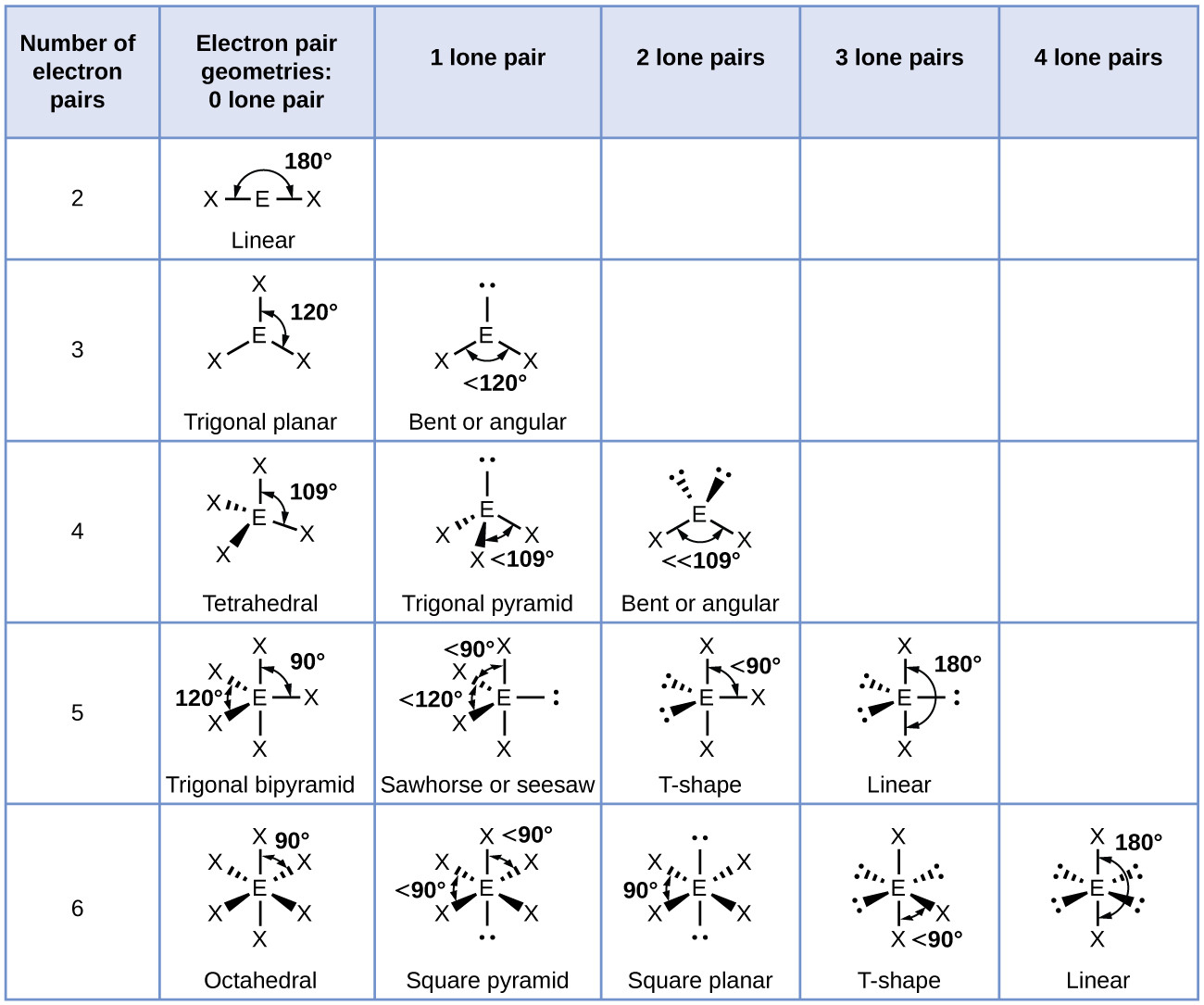
Usually, many students confuse between valence electrons and valency. The first step is - you need to calculate the number of valence electrons in the molecule. The end goal of making a Lewis structure is to identify the lone pairs of electrons and to help determine how those electrons would be bonded. When aiming to achieve the octet rule, make sure that the atom has 8 electrons in its outermost shell. Oxygen, Nitrogen, Carbon, and Halogens tend to follow this rule. The octet rule says that an atom is the most stable when it has eight electrons in its outermost shell. The Lewis structure of a compound assists in visualizing the valence electrons, and whether they exist as lone pairs or in bonds.Ī Lewis structure is drawn with the help of the octet rule. It is shown in terms of the shared pair of electrons. Lewis structure CH3OHĪ Lewis structure or an electron dot structure is a simple representation of the bonding between molecules and ions.
#MOLECULAR GEOMETRY HOW TO#
You must also know how to calculate the hybridization state of the molecule. is formed, you must know how you can draw the Lewis structure and find out the molecular geometry. It is used as a fuel and an antifreeze, in the production of hydrocarbons and solvents, and the synthesis of some other compounds such as formaldehyde. Ingesting 10ml of methanol can make you permanently blind. It is flammable and has a distinctive odor. It is a light, volatile, and colorless compound.

Methyl alcohol has characteristic properties. The general name for CH3OH is wood alcohol.

Thus, its name is methyl alcohol or methanol. The name of this chemical is derived from the common name of the alkyl group. It consists of 1 carbon atom, 3 hydrogen atoms, and 1 hydroxyl group (OH). Our study provides a fruitful avenue for applying electrostatic repulsion to modulate molecular geometry, consequently improving the optoelectronic properties of organic semiconductors.Lewis structure, Hybridization, and Molecular Geometry of CH3OH What is CH3OH?ĬH3OH is a chemical compound with the formula C H3-OH. F-2PDI-4F is one of the few examples among numerous PDI acceptors that can achieve PCEs exceeding 9%. Thus, the device with PBDB-T and F-2PDI-4F achieved a high FF of 66.9% and the best PCE of 9.05%. The performance showed that the combination of a highly crystalline donor (acceptor) and a weakly crystalline acceptor (donor) was beneficial for the improvement in FF due to the improved microstructure of the blend films. These two acceptors were incorporated with two polymer donors, PTB7-Th and PBDB-T, with varied crystallinities to fabricate a solar cell device. The repulsion of F⋯F reduced the coplanarity of PDI and created a dihedral angle of 12.6°, which endowed F-2PDI-4F with relatively weak crystallinity as compared to its analogue F-2PDI without fluorination. To tune the morphology impacted by the molecular geometry of the donor and the acceptor, a fully fused PDI derivative F-2PDI-4F, wherein the adjacent bay positions of PDI were substituted by fluorine atoms, was rationally designed and synthesized. By analyzing the photovoltaic parameters of organic solar cells based on perylene diimide (PDI) acceptors, we found that the power conversion efficiencies (PCEs) showed an approximately positive linear correlation with the fill factors (FFs), which were mainly determined by the morphology.
